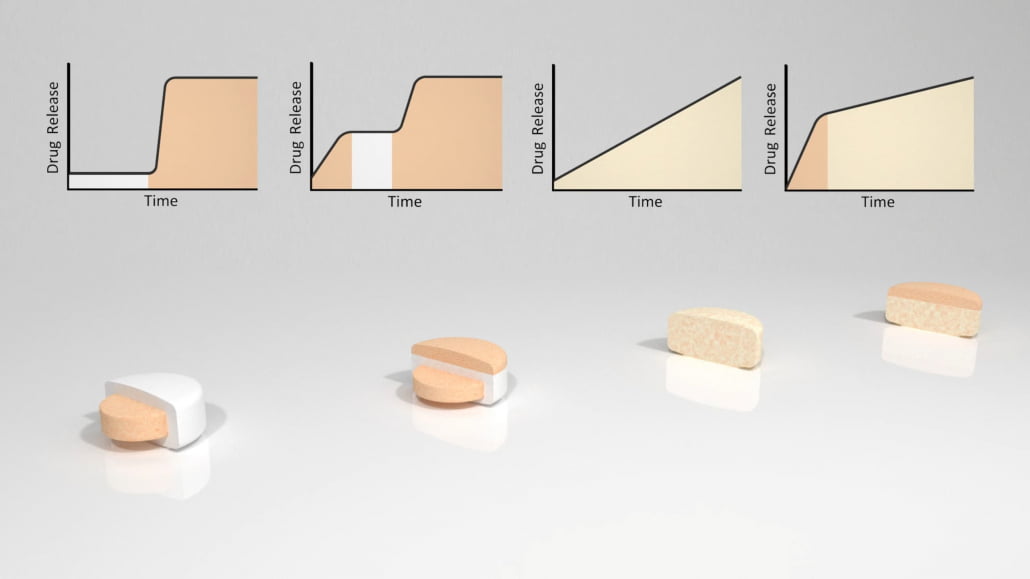
By combining our knowledge of the GI tract and the OralogiK™ technology, our formulators can target drug release to areas of the GI tract, such as the colon or upper and lower small intestine.
Reliably targeting delivery of API to the colon can be challenging due to the variable physiological environment and rates of transit along different sections of the GI tract. By applying an enteric coating around a delayed-release OralogiK™ tablet we can ensure that the erosion process only begins when the tablet has left the stomach. Knowledge of in vivo transit rates can be used to tailor the rate of OralogiK™ layer erosion to target delivery in the colon. The advantage of the erosion mechanism inherent in the barrier layer technology is that the outer layer is gradually shed from the core tablet, and on exposure of the core in the low fluid, low agitation environment of the colon, API release is relatively unimpeded by the physical presence of inert materials.
OralogiK™ has been designed to work independently of inter-individual differences such as pH, hydrodynamics and is not dependent on the microbiome for performance.